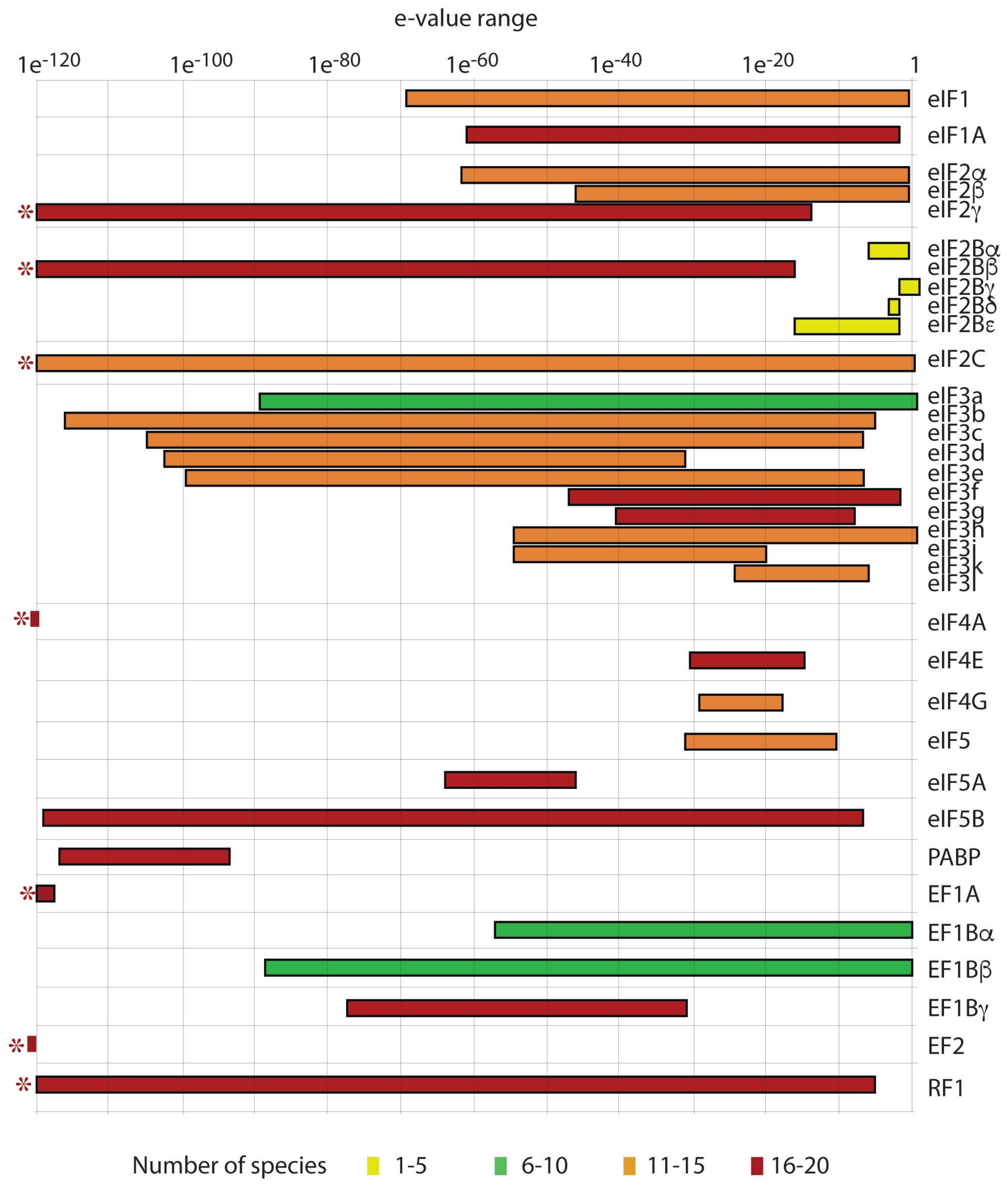
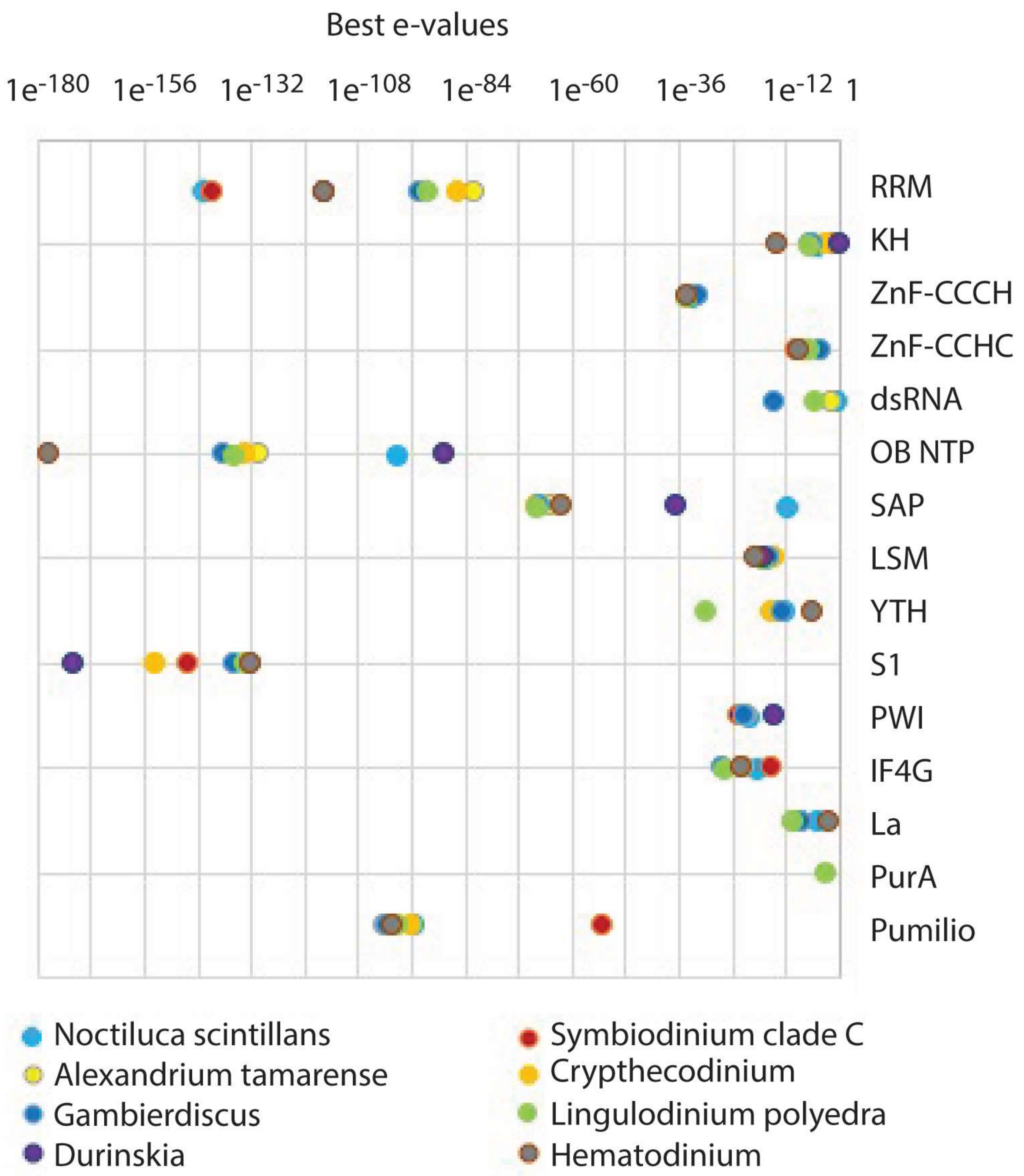
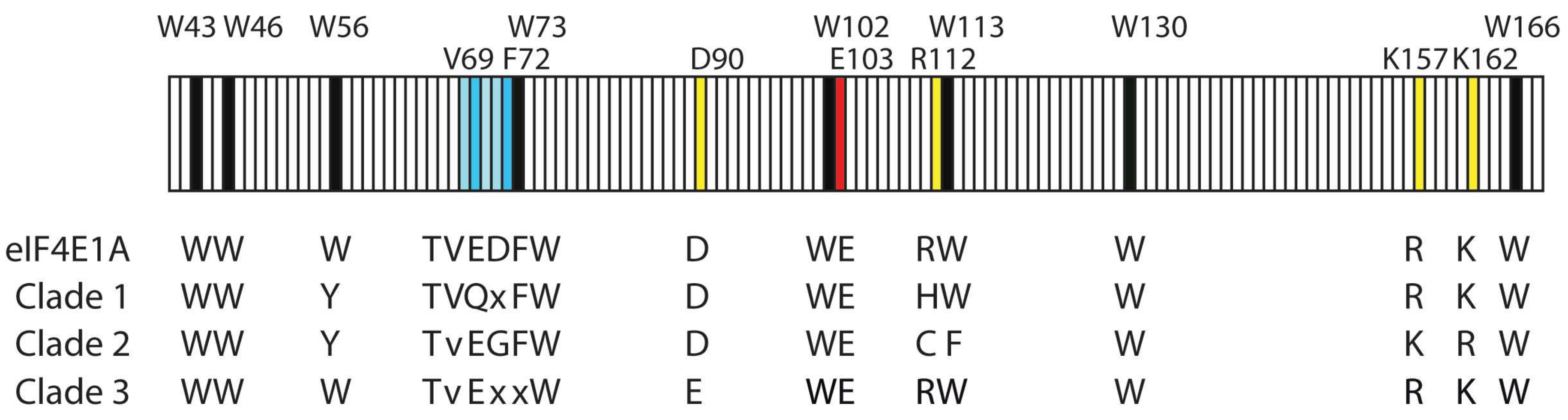
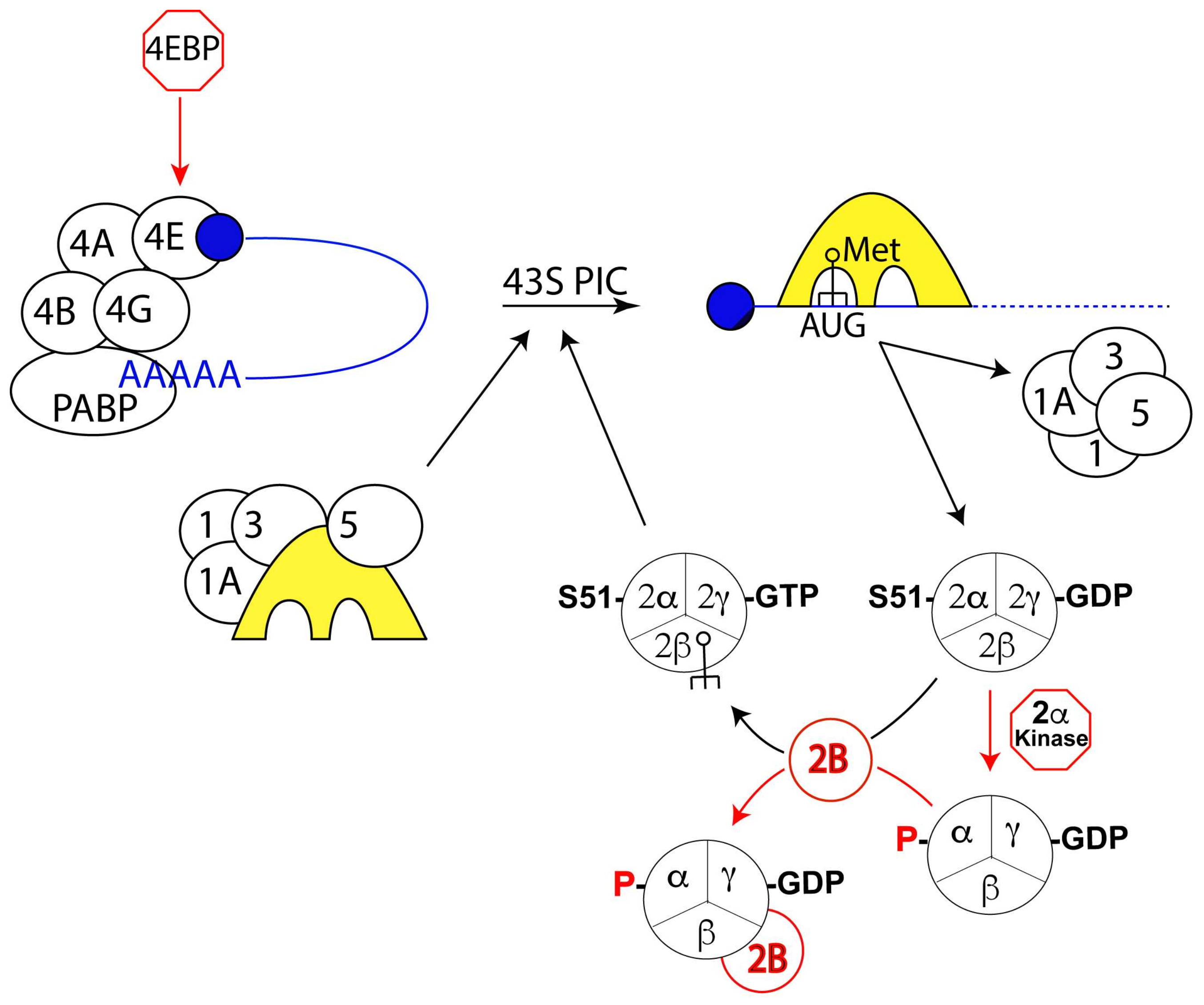
Transcriptomic, proteomic, and biochemical analyses reveal a novel neuritogenesis mechanism of Naja naja venom α‐elapitoxin post binding to TrkA receptor of rat pheochromocytoma cells - Islam - 2020 - Journal of Neurochemistry -

Human Fetal TNF-α-Cytokine-Producing CD4+ Effector Memory T Cells Promote Intestinal Development and Mediate Inflammation Early in Life - ScienceDirect

Human Fetal TNF-α-Cytokine-Producing CD4+ Effector Memory T Cells Promote Intestinal Development and Mediate Inflammation Early in Life - ScienceDirect

PDF) Phosphorylation-dependent routing of RLP44 towards brassinosteroid or phytosulfokine signalling

Human Fetal TNF-α-Cytokine-Producing CD4+ Effector Memory T Cells Promote Intestinal Development and Mediate Inflammation Early in Life - ScienceDirect

Transcriptomic, proteomic, and biochemical analyses reveal a novel neuritogenesis mechanism of Naja naja venom α‐elapitoxin post binding to TrkA receptor of rat pheochromocytoma cells - Islam - 2020 - Journal of Neurochemistry -
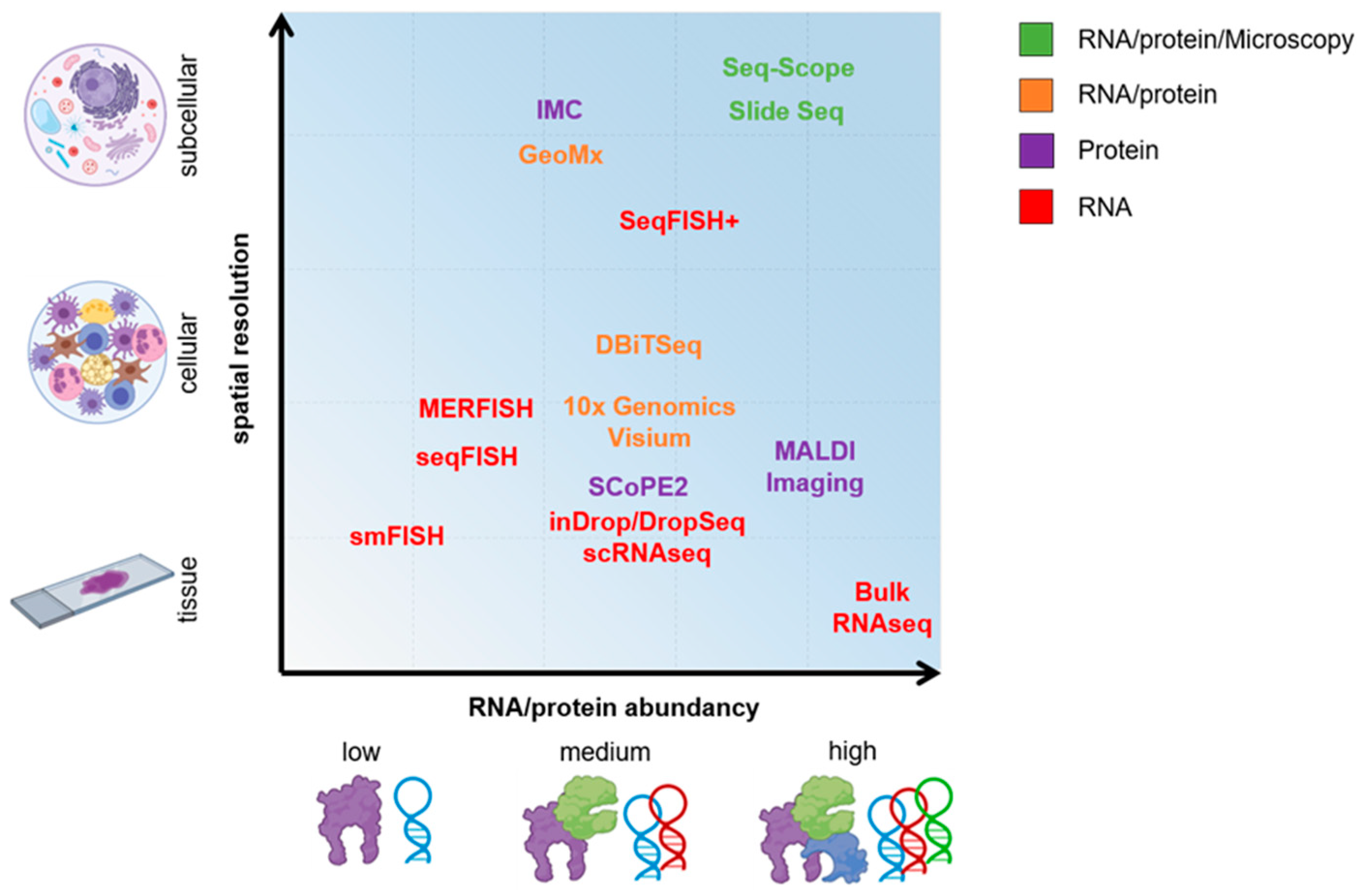
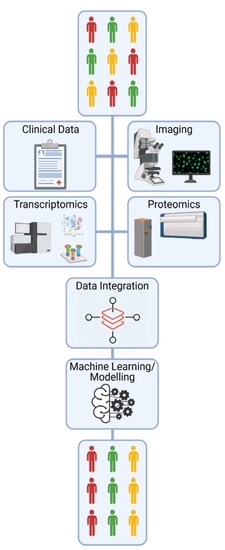
IJMS | Free Full-Text | The Potential of OMICs Technologies for the Treatment of Immune-Mediated Inflammatory Diseases | HTML